Self-described “analytics evangelist” Craig DePorter of Omya discusses why analytics enablement means learning more with less
Advocating for analytics at Omya Craig DePorter of Omya
 Meg_Hermes
Meg_Hermes
Advocating for analytics at Omya Craig DePorter of Omya
 Meg_Hermes
Meg_Hermes

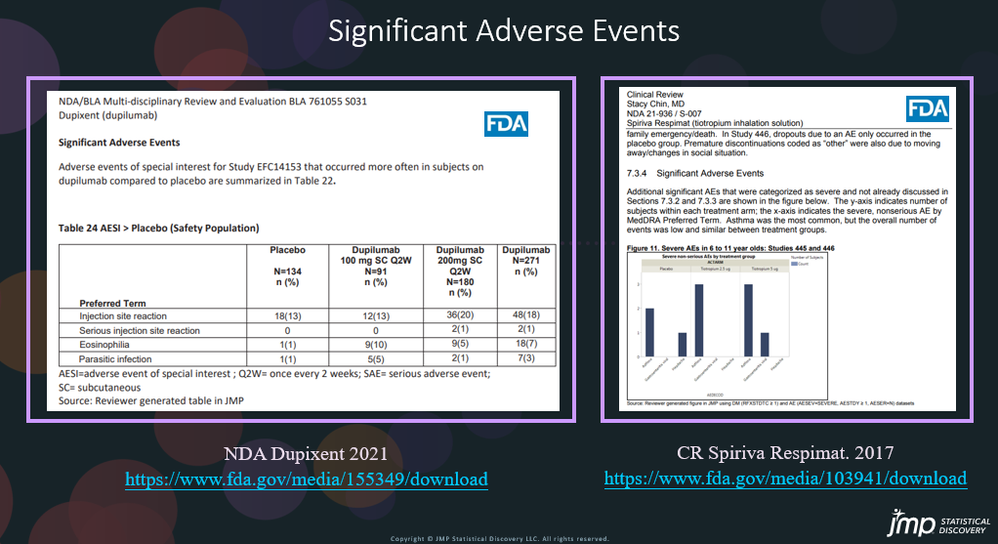
In our recent paper, CDISC Enables Efficient Streamlining of Clinical Trial Safety Evaluation1,2, we followed FDA NDA (New Drug Application) or CR (Clinical Reviews) templates to show how drug safety can be evaluated. My previous blogs demonstrated how to capture SAEs using Adverse Events Narrative3, and Discontinuations Due4 to AEs and TEAE5 with Adverse Events Distribution. There is a final step...
 Wenjun_Bao_JMP
Wenjun_Bao_JMP
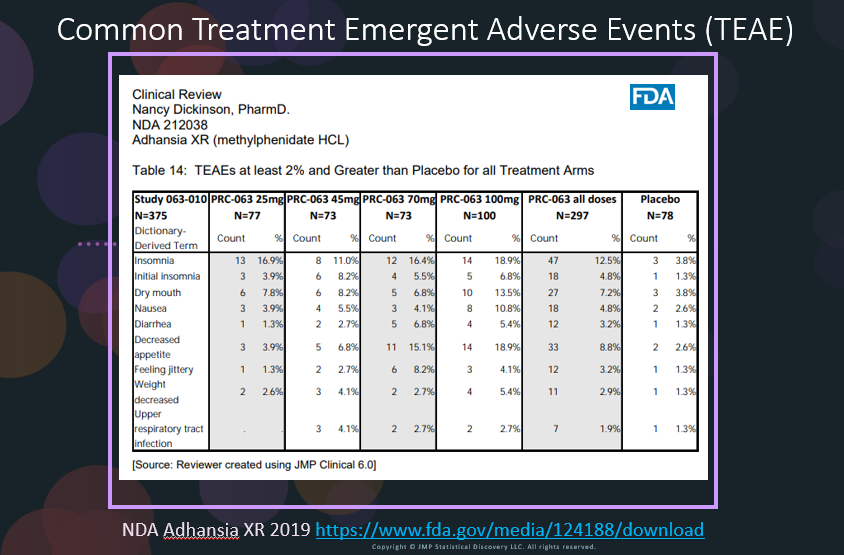
In our recent paper, CDISC Enables Efficient Streamlining of Clinical Trial Safety Evaluation1,2, we followed FDA NDA (New Drug Application) or CR (Clinical Reviews) templates to show how drug safety can be evaluated. My previous blogs demonstrated how to evaluate SAEs by Adverse Events Narrative3 and Discontinuations Due to AEs4 by Adverse Events Distribution. We'll now look at the third step for...
 Wenjun_Bao_JMP
Wenjun_Bao_JMP
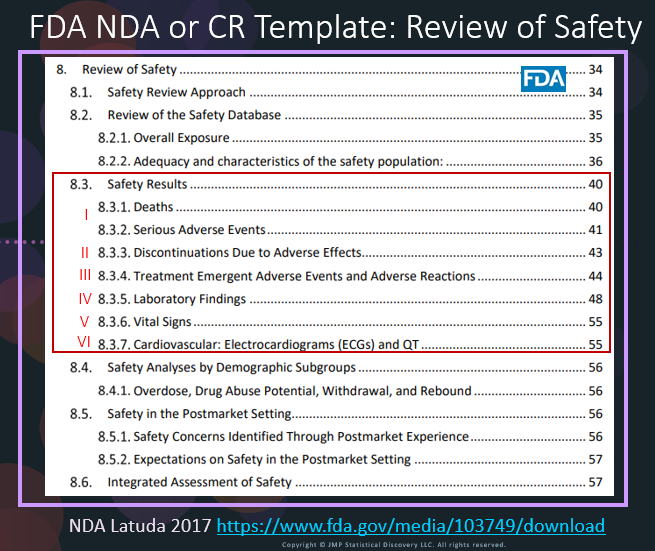
In our recent paper, CDISC Enables Efficient Streamlining of Clinical Trial Safety Evaluation1,2, we followed FDA NDA (New Drug Application) or CR (Clinical Reviews) templates to show how drug safety can be evaluated. My blog, Safety Result I, explained how to use the Adverse Events Narrative to summarize the individual information3 for those subjects that had a SAE in CR for Latuda4. The below fi...
 Wenjun_Bao_JMP
Wenjun_Bao_JMP
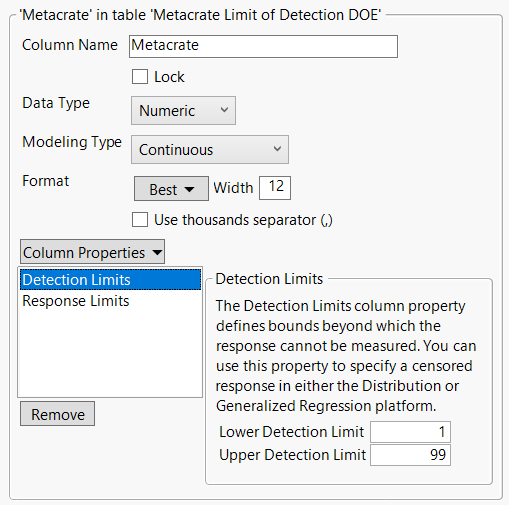
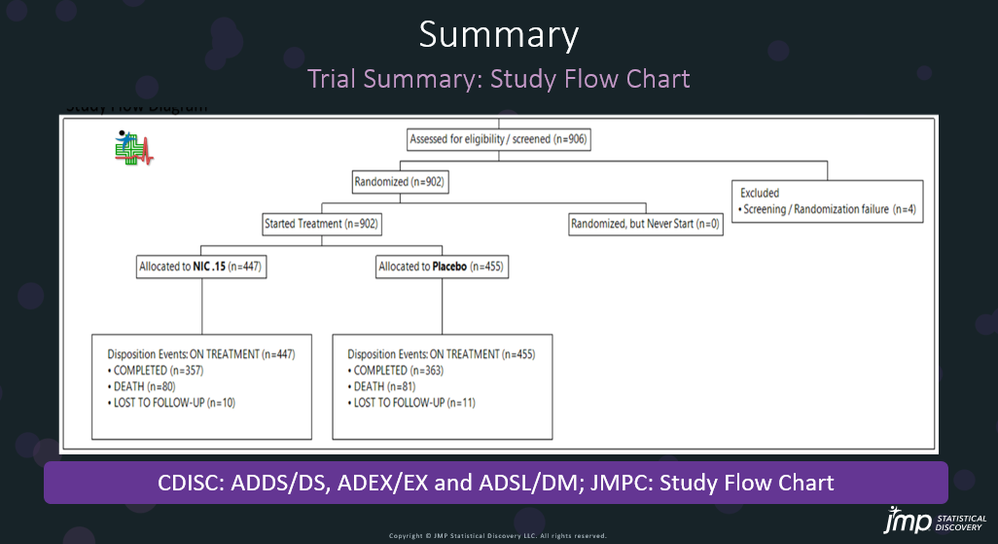
In our recent paper, CDISC Enables Efficient Streamlining of Clinical Trial Safety Evaluation1,2, we followed FDA NDA (New Drug Application) or CR (Clinical Reviews) templates to show how drug safety is evaluated by reviewers at the U.S. FDA. The typical template, shown below, is from an NDA for Mydayis3. The major content in the safety review can be found in the Safety Results section. Assessin...
 Wenjun_Bao_JMP
Wenjun_Bao_JMP

Before we can understand clinical trial data in detail, we need to understand the protocol or trial design to get a big picture of the clinical trial data. This data can be described in three ways: trial, event, and treatment emergent adverse events (TEAE) summaries, as explained in our recent paper CDISC Enables Efficient Streamlining of Clinical Trial Safety Evaluation1,2.
Trial summaries can be...
 Wenjun_Bao_JMP
Wenjun_Bao_JMP
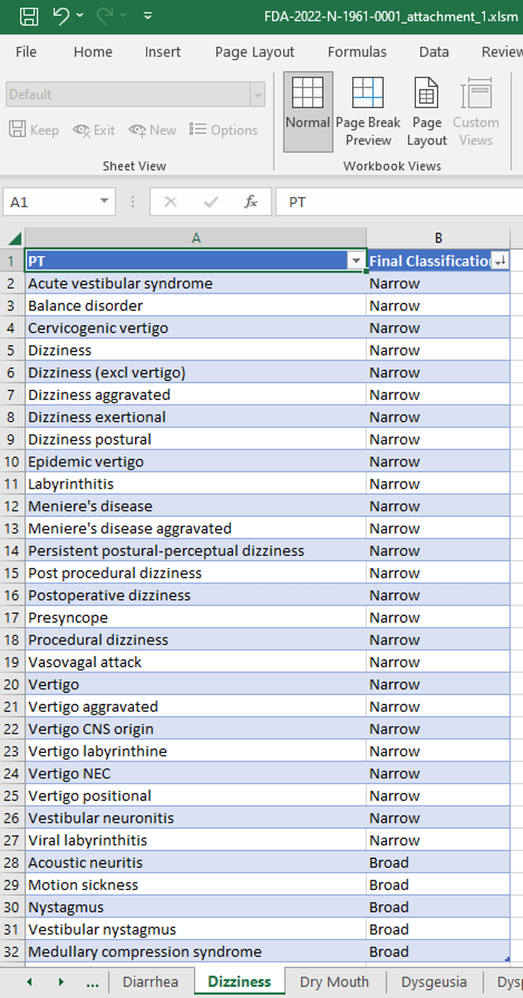
The FDA announced the release of the FDA Medical Query (FMQ) and Algorithmic FDA Medical Queries (AFMQ) at the Duke Margolis-FDA Workshop: Advancing Premarket Safety Analytics1 on Sept 14, 2022. The description of FMQ and AFMQ can be found in this FDA presentation: Advancing Premarket Safety Analytics2. Traditional medical queries (Standard Medical Queries (SMQ))3 focused primarily on MedDRA prefe...
 Wenjun_Bao_JMP
Wenjun_Bao_JMP
Creating a library of peer-reviewed articles focused on the implementation of CDISC standards.
 Wenjun_Bao_JMP
Wenjun_Bao_JMP

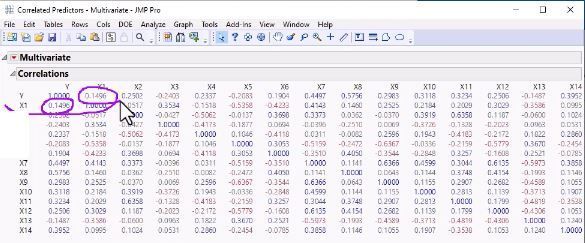
That pesky collinearity! What is it? How do I detect it? How is it related to the VIF shown in JMP reports? How do I interpret VIF?Ultimitaley, how do I build models that accurately account for collinearity? Correlation? Yes! Graph Builder Shows Negative Relationship Between Y and X2 when X1 and X2 are Both in Models
 gail_massari
gail_massari
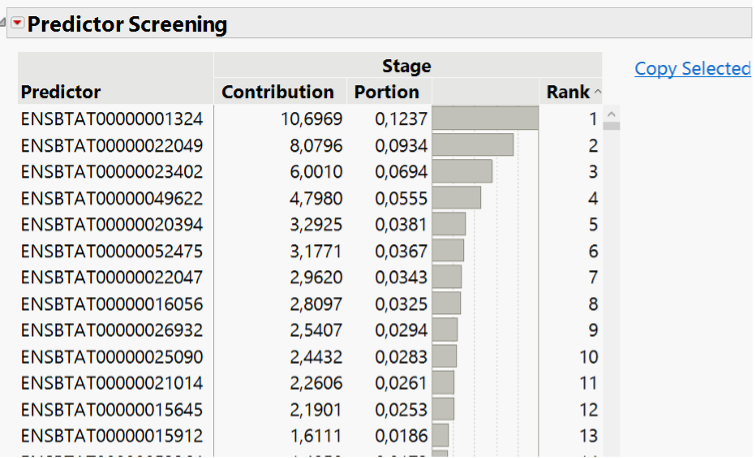
See modelization of gene expression data using JMP Pro 17.
 Valerie_Nedbal
Valerie_Nedbal
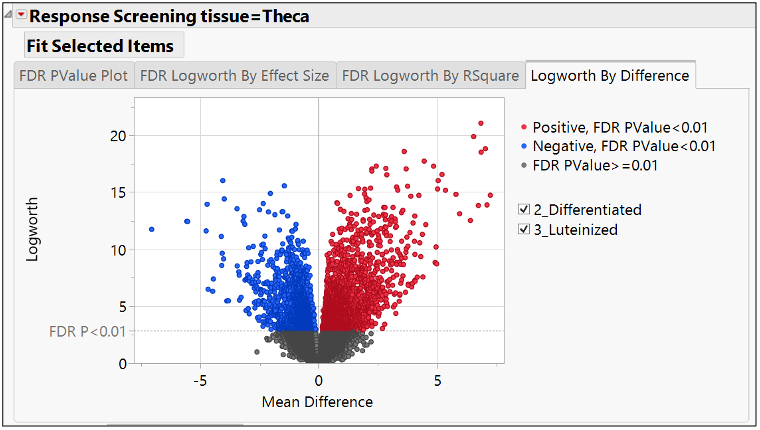
See how to compare groups for differentiated expression of genes in JMP Pro 17.
 Valerie_Nedbal
Valerie_Nedbal
JMP can connect to a number of database management systems (Oracle, SQL Server, etc.) that provide Unicode 3.5 Unicode Standard drivers. Users, new and experienced, often have data sources that require ODBC to connect JMP to a relational database to import data for analysis. Sometimes users looking to connect with their databases don't know where to begin and want to know how to obtain their datab...
 Dahlia_Watkins
Dahlia_Watkins
The "Gene Expression Data Analysis in JMP Pro" blog series continues, focused on control and quality.
 Valerie_Nedbal
Valerie_Nedbal

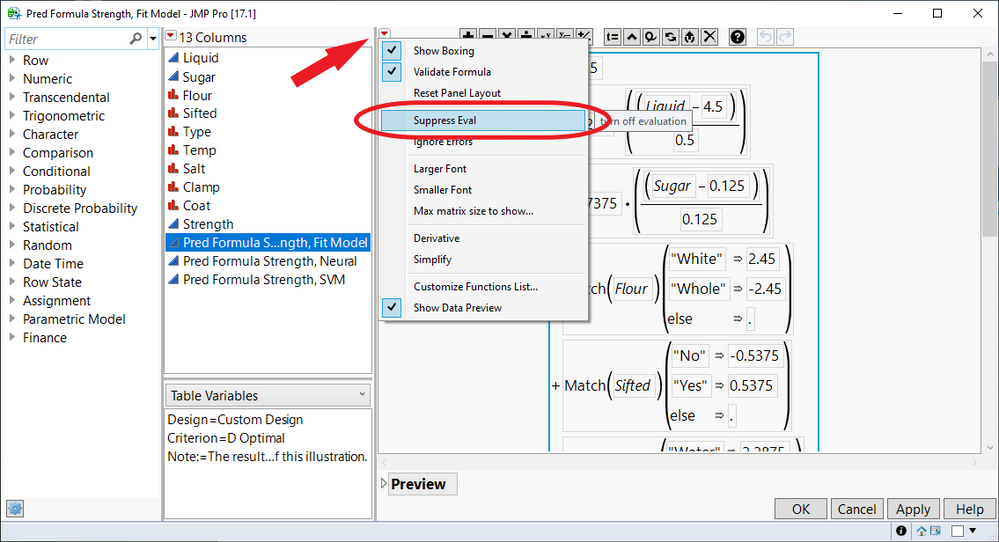
Learn when JMP suppresses formula evaluation and how you can access options to turn off suppression.
 Duane_Hayes
Duane_Hayes
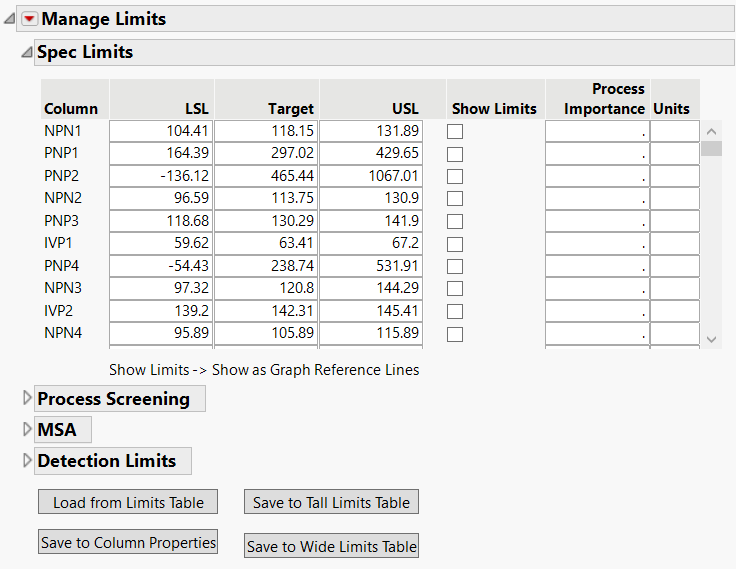
Wouldn’t it be great if the manage spec limits utility allowed you to alter more than just the specification limits? Well you’re in luck!
 tonya_mauldin
tonya_mauldin
JMP 17 brings about some key changes to the Process Screening platform. Menu changes lead to better organization and allow users to find options easier. New commands such as Filter Where, Show Charts as Selected, Chart Options, the Process Screening column property, and Platform Preferences provide key enhancements and improve the usability of the Process Screening platform.
 tonya_mauldin
tonya_mauldin

JMP 17 introduces many CUSUM (cumulative sum) control chart platform updates. New options include Save Sigma, Save Limits, Get Limits, and Platform Preferences. Also, test failures are now in the summary table.
 tonya_mauldin
tonya_mauldin

JMP 17 introduces many new updates to the EWMA (exponentially weighted moving average) platform. New options include K Sigma, Save Sigma, Connect Thru Missing, Save Limits, Get Limits, and Platform Preferences. Also, information about the average and residuals chart is now in the summary table.
 tonya_mauldin
tonya_mauldin

What if you need to pinpoint the main contributing factor to a prediction for a single person? Some modeling algorithms do not easily answer this. Some methods can. Enter Shapley Values.
 KristenBradford
KristenBradford

JMP 17 brings new features to the Distribution platform.
 tonya_mauldin
tonya_mauldin