Exploring space filling designs part 2: Comparative framework and evaluation metrics
 Victor_G
Victor_G
 Victor_G
Victor_G

 Victor_G
Victor_G

Introduction Welcome to the finale of our space filling DOE series! After establishing our framework and evaluation methodology in the previous posts, we now present the comprehensive results of our comparative study. The findings provide clear guidance on when and how to select the most appropriate space filling design for your specific application. Summary of key findings Our analysis across f...
 Victor_G
Victor_G

The Funnel Plot add-in produces funnel plots for continuous, binary, and count endpoints to assess the performance of mutually exclusive subgroups when compared to the entire population.
 Richard_Zink
Richard_Zink

Learn how JMP can be used for environmental monitoring to ensure stable processes, detect shifts early, and support regulatory compliance with real-world examples.
 Valerie_Nedbal
Valerie_Nedbal

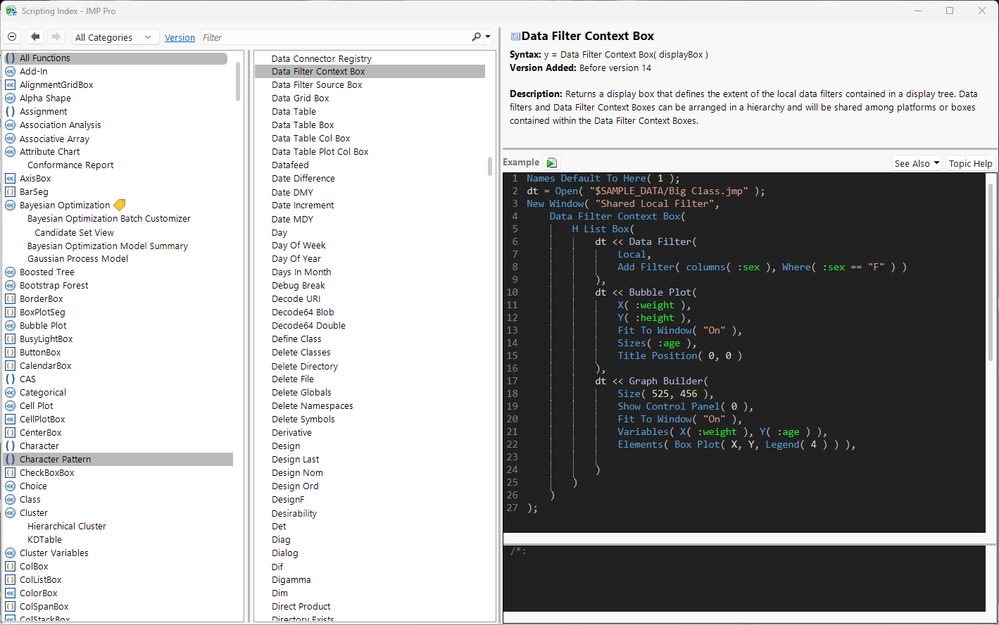
In JMP Live (and in JMP), it's possible for one local data filter to affect more than just one output in a window. Read on to learn how to use a data filter context box to make this happen.
 Jed_Campbell
Jed_Campbell
The multiple response modeling type maintains a one-row-per-patient data table while producing more informative analyses.
 Richard_Zink
Richard_Zink

Ron S. Kenett is awarded the Deming Medal and revisits the importance of information quality in this era of AI.
 anne_milley
anne_milley

I’m proposing that it’s possible to locate curvilinear ($10 word!) behavior in a designed experiment simply by using the lowly t-test.
 MikeD_Anderson
MikeD_Anderson

All a visualization really wants is to be useful.
 MikeD_Anderson
MikeD_Anderson

The visualization step is critical to doing analytics because people are truly awful at reading tables of numbers. I'm sorry, but there it is. If the table has more than a few lines, you lose almost any hope of finding trends or data quality issues. I was recently reading a book on data science for Python – guess where they started? With tables, four chapters on tables, modeling, and summary stati...
 MikeD_Anderson
MikeD_Anderson

Many people think the Distribution platform is a beginner tool or overly simplistic. That couldn't be further from the truth.
 MikeD_Anderson
MikeD_Anderson

Pythons and Pandas, Oh My!
 MikeD_Anderson
MikeD_Anderson

What if you wanted to optimize the shape of a curve? Or, understand how to control the periodicity of a response?
 MikeD_Anderson
MikeD_Anderson

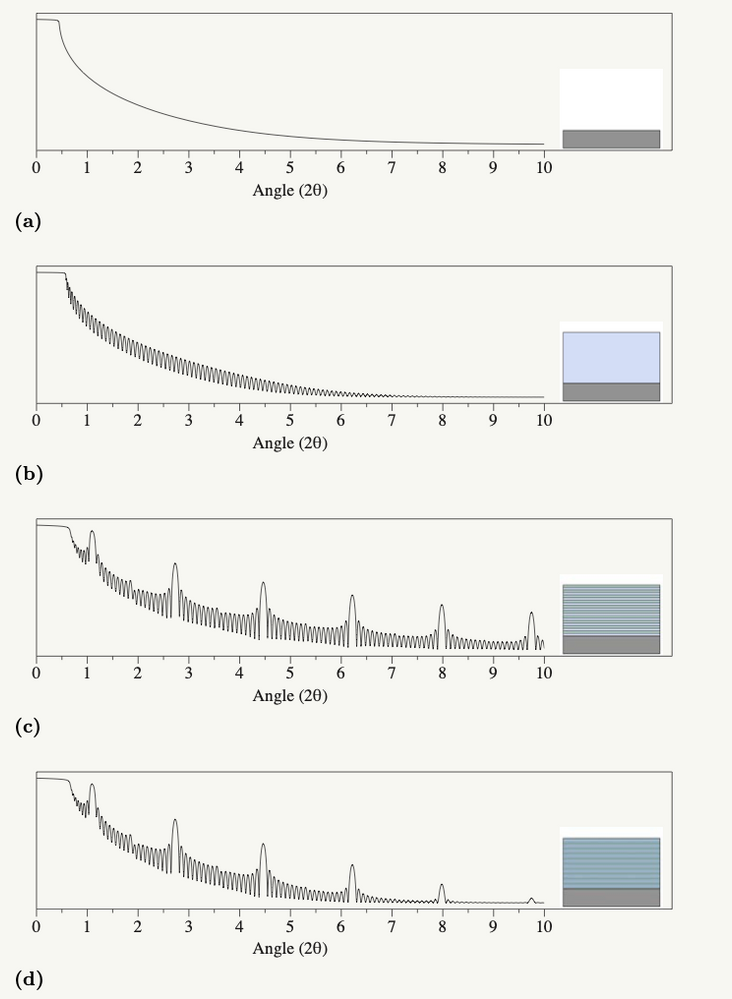
What is functional data? What does functional data analysis provide us?
 MikeD_Anderson
MikeD_Anderson

I wanted to make several graphs in the articles interactive. I knew I could create the interactive graphics in JMP and move them to JMP Public. But I didn't know how to do the bit where I embed the JMP Public content in the article. So, here is how to do it.
 MikeD_Anderson
MikeD_Anderson

JMPで同じ分析を何度も繰り返す中で、深いメニューをたどる操作に手間を感じたことはありませんか。本記事では、累積和管理図を例に、よく使う機能を素早く起動するためのショートカットキーの割り当てと、ツールバーへの登録方法を解説します。登録しておくと、今までの操作がぐっとラクになり、分析に集中できることでしょう。
 Masukawa_Nao
Masukawa_Nao

JMP is designed with a specific workflow in mind. Mastering this four-step workflow will help you to understand where to look in any JMP report for answers.
 MikeD_Anderson
MikeD_Anderson

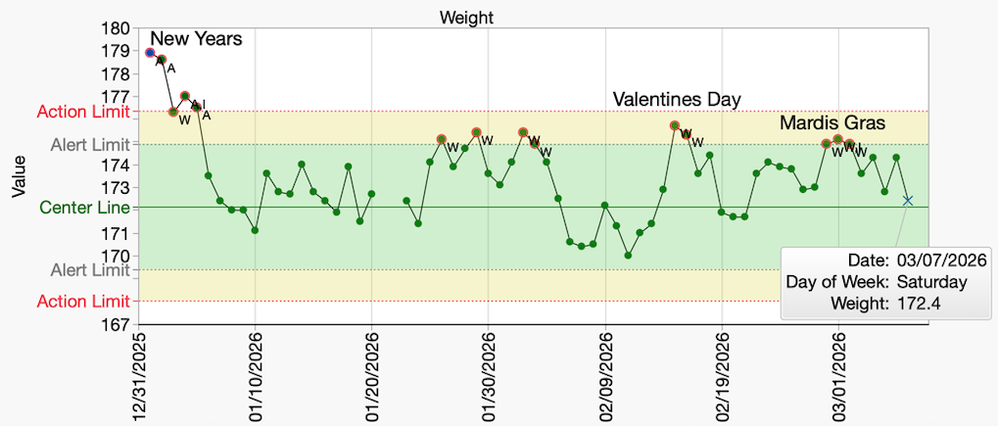
Losing weight is always a monumental task. However, maintaining your weight after the diet is over may be even harder. New JMP capabilities in JMP 19 Process Screening can help us establish expectations and provide actionable alerts to keep our weight in check!
 scwise
scwise