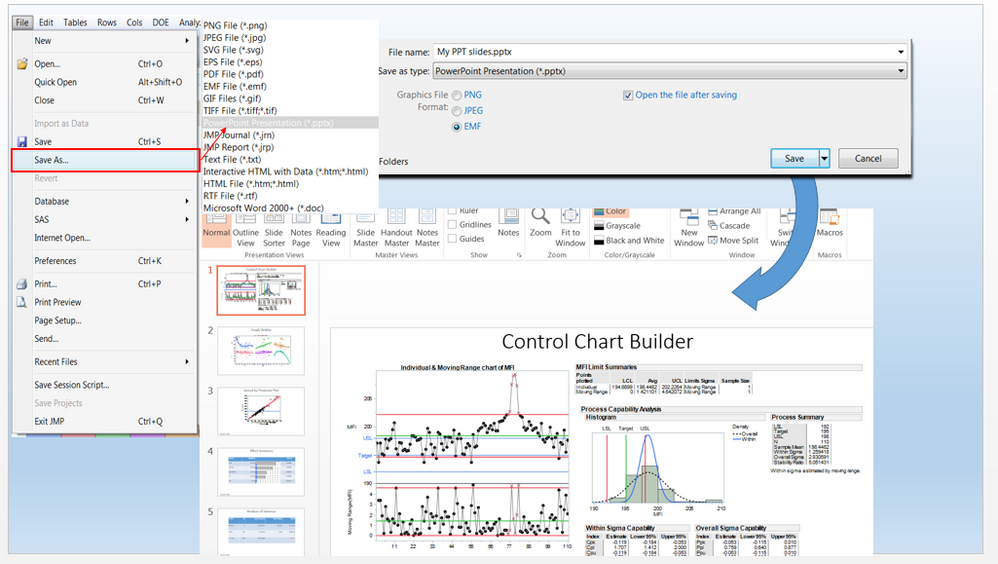
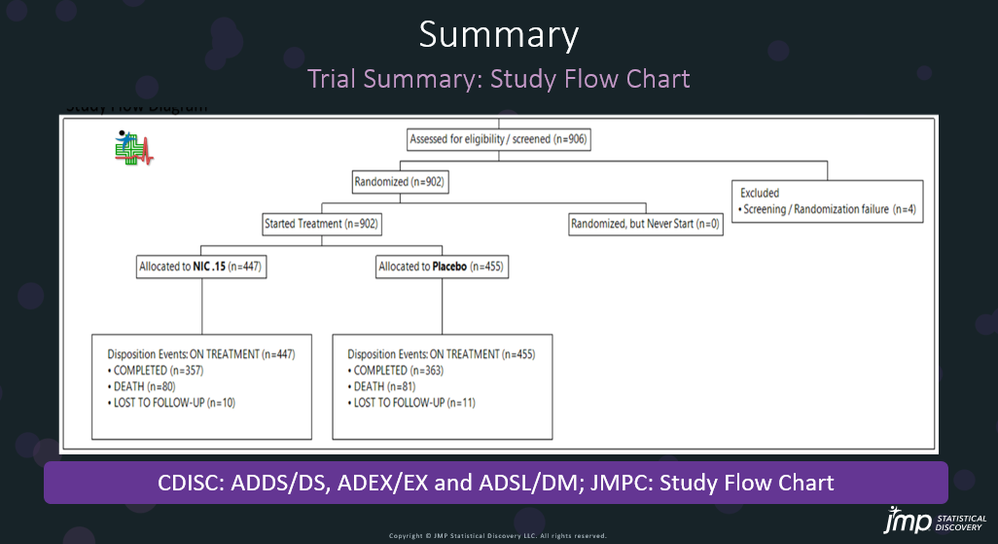
Safety Results IIIa: TEAE by Adverse Events Distribution in JMP® Clinical
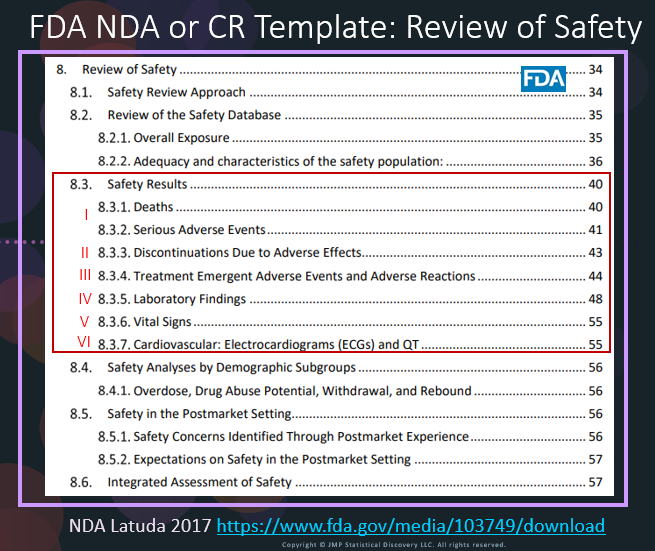
In our recent paper, CDISC Enables Efficient Streamlining of Clinical Trial Safety Evaluation 1 ,2 , we followed FDA NDA (New Drug Application) or CR (Clinical Reviews) templates to show how drug sa...
 Wenjun_Bao_JMP
Wenjun_Bao_JMP
 sseligman
sseligman

 Phil_Kay
Phil_Kay
 paul_vezzetti
paul_vezzetti
 Dahlia_Watkins
Dahlia_Watkins

 MikeD_Anderson
MikeD_Anderson
 gail_massari
gail_massari
 XanGregg
XanGregg
 Valerie_Nedbal
Valerie_Nedbal

 Duane_Hayes
Duane_Hayes
 MaryLoveless
MaryLoveless